Droplet Magnetofluidic Lamp Assay Design
Valid target sequences for primer design were first identified using the NCBI GenBank database and were checked for cross-reactivity with other organisms prior to primer design. Using PrimerExplorer V4 , several primer sets were designed against various targets in the CT genome and were tested for threshold time and absolute signal difference from baseline . Based on the results, a primer set designed to target a 321-bp region located in the CT ompA gene was selected for subsequent experiments. All primers used for this experiment are presented in Table .
LAMP reaction was then characterized for temperature sensitivity and for the effect of sample preparation reagents on amplification . For each reaction, 10L synthetic target solution was mixed with input reagent composed of 50L lysis buffer, 10L resuspension buffer, 4L magnetic particles and 30L binding buffer. Each washing step used 25L wash buffer, followed by incubation in a 25L amplification mixture.
Design And Fabrication Of Mobinaat Droplet Magnetofluidic Cartridge
The mobiNAAT cartridge was fabricated by assembling four layers of PMMA sheets as outlined in Fig. . The chamber layer was laminated with PTFE tape on one side in order to render the surface hydrophobic. The sheet was subsequently cut and laminated with the upper layer to generate hydrophilic PMMA chambers. The lower layer was laminated with PTFE film and engraved in order to expose the PMMA surface along the perimeter for bonding. A spacer frame was lined on both faces with acrylic pressure-sensitive adhesive film to provide a watertight seal and spacing between the upper and lower layers. The top layer was pre-assembled with chamber and spacer layers to form a single component, loaded with assay reagents and bonded with the lower layer. The cartridge was then flipped right side up and filled with approximately 100L FC-40 fluorinated oil. Afterwards, a mixture of 30L binding buffer and 4L magnetic particles was loaded into the sample chamber and sealed at the inlet port with plastic tape prior to transportation. Prior to each assay, sample lysate was loaded into the sample chamber of the cartridge using a disposable pipette.
Extragenital Testing Increases Case Detection Of Gonorrhea And Chlamydia: The Impact Of Implementing Nucleic Acid Amplification Testing
Dara Spatz Friedman1,2, Patrick OByrne1,2
1 Ottawa Public Health, Ottawa, ON
2 University of Ottawa, Ottawa, ON
Friedman DS, OByrne P. Extragenital testing increases case detection of gonorrhea and chlamydia: The impact of implementing nucleic acid amplification testing. Can Commun Dis Rep 2020:46:28591. https://doi.org/10.14745/ccdr.v46i09a06
Keywords: gonorrhea, chlamydia, NAAT, extragenital, pharynx, rectum
Read Also: What Medication Treats Chlamydia And Gonorrhea
Nucleic Acid Amplification Tests For The Diagnosis Of Pharyngeal And Rectal Chlamydia And Gonorrhea Infections
| The safety and scientific validity of this study is the responsibility of the study sponsor and investigators. Listing a study does not mean it has been evaluated by the U.S. Federal Government. Read our disclaimer for details. |
| First Posted : November 11, 2005Last Update Posted : September 27, 2012 |
- Study Details
| Device: Nucleic acid amplification test | Not Applicable |
In the latter part of the 1990’s, testing for Chlamydia trachomatis and Neisseria gonorrhoeae infections was revolutionized by the introduction of nucleic acid amplification tests which achieve greater sensitivity than traditional culture methods by exponentially replicating the nucleic acid of these organisms. The specimens collected for NAATs are easier to transport and NAATs permit collection of less invasive specimens for testing . Despite their somewhat higher costs, these tests have been found to be preferred by patients , by clinicians , and to permit expanded screening both in traditional clinical settings and at outreach sites where testing has not typically been done in the past.
Exclusion Criteria:
- History of receipt of antimicrobial agents active against N. gonorrhoeae or C. trachomatis in the preceding 30 days.
What Happens During A Gonorrhea Test
If you are a woman, a sample may be taken from your cervix. For this procedure, you will lie on your back on an exam table, with your knees bent. You will rest your feet in supports called stirrups. Your health care provider will use a plastic or metal instrument called a speculum to open the vagina, so the cervix can be seen. Your provider will then use a soft brush or plastic spatula to collect the sample.
If you are a man, your provider may take a swab from the opening of your urethra.
For both men and women, a sample may be taken from a suspected area of infection, such as the mouth or rectum. Urine tests are also used for both men and women.
Some gonorrhea tests can be done with an at-home STD test kit. If your health care provider recommends at-home testing, be sure to follow all directions carefully.
Your health care provider may order tests for other STDs when you get a gonorrhea test. These may include tests for chlamydia, syphilis, and/or HIV.
Don’t Miss: How Long Can You Leave Chlamydia Untreated
Patient And Public Involvement
Vaginal specimens are an acceptable and sometimes a preferred way for chlamydia and gonorrhoea testing among women. In this study, we compare the evidence base of performance of different testing sites for detection of chlamydia and gonorrhoea by conducting a systematic review of published primary research. We did not involve patient groups in the design or conduct of the study. The findings of the study can be used to standardise testing practices.
Questions For Your Doctor About Test Results
It can be helpful to bring questions to your doctor to learn more about your chlamydia test results. Helpful questions may include:
- What is my chlamydia test result?
- Did my test check for any other STDs?
- Do I need any treatment based on my results?
- How can I talk to my sexual partners about chlamydia?
- When should I be tested for STDs and how often?
Also Check: Can Boric Acid Cure Chlamydia
Submission And Collection Notes
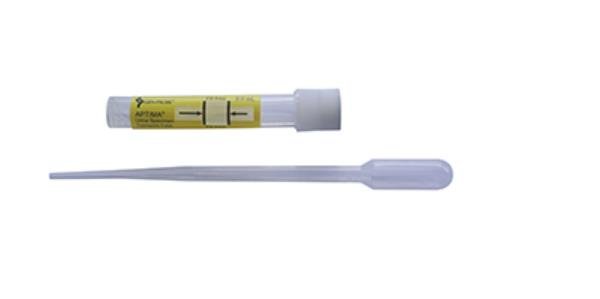
Collect 20-30 ml of urine in a sterile container, then transfer 2 ml of urine from the sterile container to the Hologic® Aptima® Urine Transport tube between the black fill lines using the disposable pipette within 24 hours of collection. For details on specimen collection refer to the instructions located on the Hologic® Aptima® Urine Specimen Collection kit packaging.
The Hologic® Aptima® Urine Transport tube Urine collection kits contain a preservative the transferred urine must be between the black fill lines for accurate testing.
Neat urines, and urine submitted in a kit other than the Hologic® Aptima® kit will be rejected. The Hologic® Aptima® Urine Specimen Collection kit is the only acceptable collection kit for testing.
The patients date of birth must be indicated on the in order for the specimen to be processed for testing.
Appendix: Key Questions Reviewed By The Work Group And Member Responsible For Literature Summary
Performance Characteristics
- Does the sensitivity and specificity of available tests for C. trachomatis and N. gonorrhoeae vary with respect to anatomic site from which the specimen was collected and/or specimen type?
- Joan Chow, DrPH, California Department of Public Health, Richmond, California
- Katherine Whitaker, PhD, Food and Drug Administration, Rockville, Maryland
Screening Applications
- What specimen types are optimal for of C. trachomatis and N. gonorrhoeae screening purposes?
- Charlotte Gaydos, DrPH, Johns Hopkins University, Baltimore, Maryland
- Sarah Guerry, MD, Los Angeles County Department of Health Services, Los Angeles, California
- Barbara Van Der Pol, PhD, Indiana University School of Public Health, Bloomington, Indiana
Economic Considerations
You May Like: Does Chlamydia Ever Go Away
Example Of This Test In Action
NAATs are incredibly sensitive methods of detecting whether a bacteria or virus is present in a biological sample. When it comes to detecting genital herpes in a sore from a person who has symptoms, these tests serve as a viable alternative to viral culture. Viral cultures can be difficult for some laboratories to perform.
Unlike herpes blood tests, a NAAT still involves the direct determination of whether a virus is present in the sample rather than looking for anti-herpes antibodies.
Nucleic-acid amplification has also allowed for an expansion of chlamydia and gonorrhea screening around the country. Now such screening can now be done on urine samples instead of requiring a urethral or cervicovaginal swab. It has thus become easy to test large numbers of young men and women for these STDs in a variety of both clinical and non-clinical settings. Collecting urine requires no medical expertise. People are also more likely to be willing to pee in a cup than undergo a genital swab.
Researchers have also used NAATs to get more information about the extent of the problem of asymptomatic STDs in the United States. Large-scale NAAT-based screening programs have been implemented in the military, in urban teenagers, in men who have sex with men, and in other high-risk and low-risk groups. These tests allow for the detection of STDs in the small urine or blood samples that are often taken as part of large research studies on population health.
When Should I Get Chlamydia Testing
As most people infected with chlamydia do not experience symptoms, doctors rely on screening to detect most cases of chlamydia. Screening guidelines vary based on many factors, including a persons anatomy, health, and sexual practices. Regular screening for chlamydia is recommended for several groups:
Certain factors increase the risk of contracting chlamydia and may affect how often a person should be screened. Risk factors include having:
- Sex with a new partner
- More than one sexual partner or a partner who has sex with mutiple people
- A sex partner diagnosed with an STD
Testing for chlamydia is more frequently conducted in asymptomatic people in settings where infection rates are high, which often includes correctional facilities, adolescent health clinics, the military, and sexual health clinics.
Diagnostic chlamydia testing is recommended for anyone with signs or symptoms of this infection. When symptoms do occur, they may not appear until a few weeks after exposure. Signs and symptoms of chlamydia can vary based on the site of infection but may include:
- Burning during urination
- Abnormal discharge from the vagina, penis, or rectum
- Vaginal bleeding after sex or pain during intercourse
- Pain, tenderness, or swelling in the testicles or scrotum
- Rectal pain
Also Check: Is It Possible To Not Pass Chlamydia
Design And Fabrication Of The Mobinaat Cartridge
Computer-assisted drawing software was used to construct a prototype enclosure that incorporates an Arduino Uno R3 microcontroller and motor shield, a rotary actuator , thermal incubators and optical components . All components were designed and assembled using PMMA sheets and acrylic adhesive solvent where necessary. A pair of cylindrical NdFeB magnets was affixed on each of the spoke tips to provide permanent magnetic fields for particle manipulation.
The software for the mobile app was developed using Android Studio . In relation to the processing module, the app operates as a serial communicator that initiates various pre-programmed routines on the microcontroller via Bluetooth communication. The mobile app was designed to provide three components: a training module for naïve operators main routine which operates the processing module and performs data acquisition data viewer with signal processing algorithm to differentiate positive from negative samples. A Samsung Galaxy S3 was used as the mobile device for this study.
When Should I Get A Gonorrhea Test
Because many people who are infected with gonorrhea do not have any noticeable symptoms, a number of health organizations recommend regular gonorrhea screening for certain adolescents and adults between the ages of 15 and 65, including:
- Women under 25: All sexually active women younger than age 25 should get yearly screening for gonorrhea.
- Women 25 and older: Women 25 and older who have risk factors for gonorrhea infection should be screened yearly.
- Pregnant women: Because gonorrhea can be passed from mother to child during childbirth, all pregnant women under 25 should be tested. Pregnant women 25 and older should be screened if at an increased risk of gonorrhea infection.
- Men who have sex with men: Men who have sex with men should be screened annually for gonorrhea, or every 3 to 6 months if they are at an increased risk for contracting gonorrhea.
- People with HIV: People diagnosed with HIV should be screened for gonorrhea at least once per year.
Regular screening for gonorrhea in heterosexual men without symptoms is not generally recommended.
Risk factors that impact the frequency of gonorrhea screening include:
- A new sexual partner in the past 60 days
- Multiple sex partners
- Painful bowel movements
- Sore throat
After being treated for gonorrhea, its recommended to test for gonorrhea again three months after completing antibiotics.
Don’t Miss: How Long Before Chlamydia Turns Into Hiv
Specimen Collection And Handling
Urine specimens are the specimen of choice for males and post-hysterectomy female patients.
For females in general, urine is a second-line option, because it is less sensitive than cervical swabs for CT/GC NAAT. For testing non-post-hysterectomy female patients, please refer to the test information sheet for specimen collection and testing information.
Summary Of Recommendations And Evidence
The USPSTF recommends screening for chlamydia in sexually active women aged 24 years or younger and in older women who are at increased risk for infection . B recommendation.
Screening for Chlamydia and Gonorrhea: Clinical Summary of the USPSTF Recommendation
Population
note: For a summary of the evidence systematically reviewed in making this recommendation, the full recommendation statement, and supporting documents, go to .
HIV = human immunodeficiency virus STI = sexually transmitted infection USPSTF = U.S. Preventive Services Task Force.
Screening for Chlamydia and Gonorrhea: Clinical Summary of the USPSTF Recommendation
Population
note: For a summary of the evidence systematically reviewed in making this recommendation, the full recommendation statement, and supporting documents, go to .
HIV = human immunodeficiency virus STI = sexually transmitted infection USPSTF = U.S. Preventive Services Task Force.
The USPSTF recommends screening for gonorrhea in sexually active women aged 24 years or younger and in older women who are at increased risk for infection. B recommendation.
See the Clinical Considerations section for a description of populations at increased risk for infection.
The USPSTF concludes that the current evidence is insufficient to assess the balance of benefits and harms of screening for chlamydia and gonorrhea in men. I statement.
Recommended Reading: How Do Men Test For Chlamydia
Sample Collection Handling And Shipment
Aptima® Urine Kit for Urine specimens only Aptima® Unisex Swab Kit for Endocervical and Male Urethral Swab Specimens Aptima® Multitest Swab Kit for patient-collected vaginal swabs, oral swabs, and rectal swabs
Assay Workflow Design For Mobinaat Platform
The mobiNAAT platform workflow begins with a two-step sample loading, where the sample matrix presented in the form of a vaginal swab is initially expressed in a tube containing lysis reagent for cell lysis. When the expressed sample is transferred from the tube into the cartridge using a syringe, the sample can be further lysed by attaching a syringe-linked bead-beating module for mechanical disruption of remaining cells. With the sample loaded, the cartridge is inserted into the cartridge-processing unit. The operator is assisted by a checklist on the mobile app to ensure that the manual steps have been performed correctly. Each step of the assay is clarified further by the inclusion of a video instruction embedded in the app . Once all of the manual steps are completed in approximately 2minutes, the mobile phone is docked into the cartridge-processing unit, and automated cartridge processing is initiated.
Figure 2
In order to assess the accessibility of the platform to a context-relevant end user, an on-site nontechnical research coordinator using the platform for the first time was trained using the training modules accessed via the mobile app. Subsequent comparison of results obtained from the app-trained naïve operator and platform developers showed full correspondence for the samples tested, demonstrating the ease of use facilitated by the mobile phone interface and the training module .
Also Check: What Medicine Is Used To Treat Gonorrhea And Chlamydia
Samples Will Be Rejected If They Are:
- Unlabeled specimens or those with discrepancies between specimen and requisition forms All specimens MUST have a unique patient identifier B barcode from LIMS.
- Insufficient in Quantity No specimen received, no specimen in container, or insufficient specimen to perform testing. Specimens received with no orders or requisition forms.
- Improperly Preserved Specimens must be refrigerated or stored at room temp for 30 days as defined by the laboratory. Tubes have collection media in the container that must be maintained in the tube with the swab. Expired transport containers, specimens received after prolonged delay or inappropriate transport conditions.
- Damaged Specimen leaked or broken in transit, dry specimens, or in fixative or other additive.
- Wrong swab the collection kit has a white cleaning swab and blue collection swab, only the blue swab can be used for testing. Samples submitted in bacterial culturettes, multiple swabs, white cleaning swab, wrong transport tube, or other collection containers or swabs.
- Overfilled or Underfilled Urines The yellow top tubes have a window where the urine volume should be filled to those over or under this level will be rejected.
Return to the Delaware Public Health Laboratory page